This session is going to be an in-depth look at fluids, so let’s learn more about them in detail. Let’s learn about the different properties of fluids, their characteristics, definitions, and examples that are relevant to our daily lives.
What is Fluid?
Fluids constantly deform, so they flow without interruption. Fluid is a substance that has no shape and easily yields to pressure from the outside. At rest, fluids are liquids, gases, or any material that cannot withstand forces such as shearing or tangential forces.
When these forces are applied to the fluid, it undergoes a continuous change in shape. Fluids are substances with zero shear modulus, meaning they are unable to withstand shear forces.
In daily life, we would have heard the dehydrated patient instructed to drink more liquids to compensate for the water loss. We should drink more fluids because they aid in the proper digestion of food.
You might like: Different Types of Plumbing Tools [Names & Pictures]
Characteristics of Fluids
According to their flow, fluids are divided into four types, which are as follows:
- Steady or unsteady
- Compressible or incompressible
- Viscous or non-viscous
- Rotational or irrotational
#1 Steady or Unsteady
It is the fluid that, while flowing, maintains a constant density throughout. While flowing, the unsteady fluid’s velocity varies between any two points.
#2 Compressible or Incompressible
These fluids are categorized according to their Mach Number. The compressible fluid has a Mach Number between 0.3 and 1, while the incompressible fluid has a Mach Number of 0.3.
#3 Viscous or Non-Viscous
Viscous fluids are those that are thicker or more viscous; they are typically very gloppy fluids. Motor oil and shampoo are two examples.
Non-viscous fluids are defined as those fluids that are relatively thinner or less viscous. These fluids have no or very little resistance to internal friction. Fluids that are non-viscous move without losing any kinetic energy. For Instance: Superfluid liquid helium.
#4 Rotational or Irrotational
In rotational flows, the angle between intersecting lines of the fluid element’s boundary changes as it moves in the flow. The fluid is divided into rotational fluid and irrotational fluid based on the angular motion.
Irrotational flow is characterized by fluid rotation without any change in angle between the boundary lines. These fluids are classified into two types based on their research:
- Fluids statics: It is the mechanism of fluids in motion or at rest as well as the pressure that fluids place on anyone.
- Fluids dynamics: It includes the study of fluid motion and flow. Fluid dynamics includes well-known subfields like aerodynamics and hydrodynamics.
You might like: What are Flow Control Valves? [PDF]
Properties of Fluids
An understanding of these properties is necessary for us to implement the basic principle of fluid mechanics in the solution of practical problems.
The following are the different properties of fluids:
- Density or Mass density of the fluid
- Specific weight or weight density of the fluid
- Specific Volume
- Compressibility
- Specific gravity
- Surface tension
- Capillarity
- Viscosity
- Temperature
- Pressure
- Cavitation

Read Also: What are the different types of flow control valves?
#1 Density or Mass Density of the Fluid
The mass density of the fluid is described as the ratio of the mass of fluid to its volume. The density of the fluid can also be defined as the mass per unit volume of the fluid. The density of the fluid is denoted by the symbol ρ (rho).
In the SI system, the unit of measurement of the mass of the fluid is kg and that of the volume of fluid is cubic meter m3. Thus the unit of measurement of density is kg/cubic meter (kg/ m3). The density of the liquids is considered to be constant with the temperature but of the gases, it changes with the temperature.
The density of water is considered to be 1000kg/ m3 or 1gm/cm3 and it is standard for the measurement of the density of the other fluids.
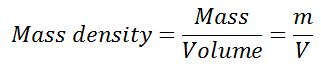
Read also: Define Fluid and Types of Fluids in Fluid Mechanics
#2 Specific Weight or Weight Density of the fluid
It is defined as the ratio of the weight of the fluid to the volume of fluid. It is also defined as the weight of the fluid per unit volume of fluid. The weighted density of the fluid or specific weight is indicated by the letter w.
In the SI system, the unit of measurement of the weight of the fluid is N (Newton) and the volume is cubic meter m3, so the unit of measurement of specific weight is N/m3 “Newton per meter cube”. The value of the specific density of water is 9.81×1000 Newton/m3 in an SI unit.
The relationship between g and can be determined by Newton’s 2nd Law since weight per unit volume = mass per unit volume x g
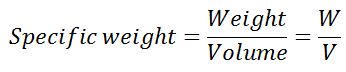
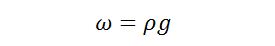
#3 Specific Volume
The specific volume of the fluid is occupied by the unit mass of the fluid. It is also defined as the volume per unit mass of the fluid.
The specific volume of the fluid is reciprocal to the density of the fluid so its unit of measurement in the SI system is m3/kg. This item is important for the gases.
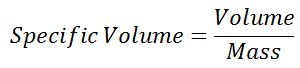
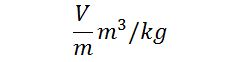
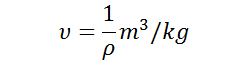
Read also: Difference Between Hydraulic and Pneumatic
#4 Compressibility
It is the reciprocal of the bulk modulus of elasticity, the ratio of compressive stress to volumetric strain defined as K.
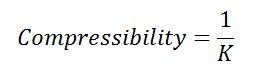
Where bulk modulus is the ratio of volumetric stress and volumetric strain,
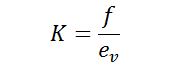
#5 Specific Gravity
The specific gravity of the fluid is defined as the ratio of the density of the fluid to the density of the standard fluid. For liquids the standard fluid is water and for gases it is air.
It is also called relative density. Since specific density is the ratio of the two densities, it is a dimensionless quantity and has no unit of measurement. It is denoted by the symbol S.
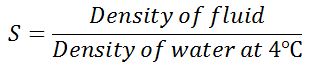
Read Also: What are the Types of Fasteners? Their Uses & Examples [PDF]
#6 Surface Tension
A liquid creates an interface with the second liquid or gas. The surface energy per unit area of the interface is known as the surface tension coefficient of surface tension. It is also described as the tensile force on the surface of the liquid in contact with a gas, such that the contact surface acts as a membrane under tension.
Liquids have both cohesion and adhesion and both are forms of molecular attraction.
- Cohesion allows a liquid to resist tensile stress
- Adhesion allows it to adhere to another body.
At the interface between a liquid and a gas, that is, in a liquid, the out-of-equilibrium force between molecules creates an imaginary surface layer, which exerts a tension force on the surface. This liquid property is known as surface tension.
Surface tension has the unit of force/unit length or energy/unit area. Surface tension = Force per unit length in N/m.
The most common interfaces and values of S for the clean surface at 20°
= 0.073N/m for air-water surface
= 0.48 N/m for air mercury interface
#7 Capillarity
The rise or fall of liquid in small diameter tubes is due to molecular attraction is called capillarity.
Liquids, such as water, which wet a surface cause capillarity rise. In non-wetting liquids (mercury) capillarity depression is caused.
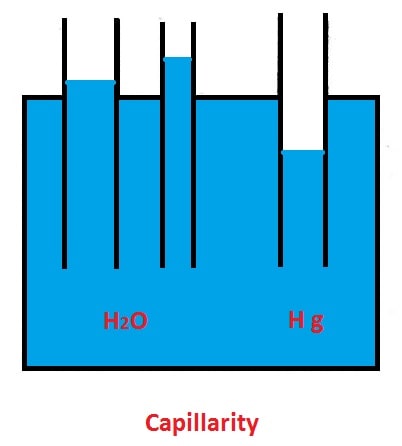
Surface tension occurs during the gas-liquid interface, but if that interface comes into contact with a solid surface like the walls of a container, the interface usually decreases near or below that surface. The shape of such a concave or convex surface is known as the meniscus.
#8 Viscosity
Liquids such as water or oil flow as layers on the surface, with the top layer moving at the fastest speed and the bottom layer moving at a slower speed. This shows that as it moves from the top layer to the bottom layer the speed of the flow of fluid reduces.
There is some sort of resistance to the flow of various layers of the fluid, which is offered by the adjoining layers, this property of the fluid is called the viscosity of the fluid. The viscosity of the fluid is defined as the property of the fluid that offers resistance to the movement of one layer of the fluid over another adjacent layer of the fluid.
It is the resisting property of a fluid-to-shear force. Viscosity, i.e., is the property of a fluid, due to cohesion and interaction between molecules, which offers resistance to shear deformation. Under the same shear stress, different fluids deform at different rates.
The viscosity of the fluid is Ns/ m2 in the SI unit of measurement
A fluid with high viscosity such as syrup deforms more slowly than a fluid with low viscosity such as water. All fluids are viscous “Newtonian Fluids” follow the linear relation given by Newton’s law of viscosity.
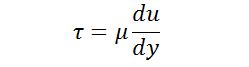
Where 𝜏 is the shear stress, Units is N/ m2; kg/ ms2
du/dy is a velocity angle or rate of the shear strain it has units: radians.
μ is the “coefficient of dynamic viscosity”.
#9 Temperature
The body’s level of heat or cold is influenced by its temperature. The fluid’s temperature can be expressed in Kelvin, Celsius, or Fahrenheit. The Kelvin scale is used according to the International System of Units.
#10 Pressure
A fluid’s pressure is defined as the force it exerts per unit of area. There are two situations in which fluid pressure occurs: an open situation and a closed situation.
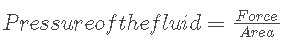
#11 Cavitation
Cavitation is the formation of bubbles or voids within a fluid. This happens when the pressure drops below the vapour pressure in an instant.
Conclusion
That’s it thanks for reading. If you like our article on “Properties of Fluids” please share it with your friends. If you have any questions about the properties of fluids just leave a comment I’ll respond to you.
Want free PDFs direct to your inbox? Then subscribe to our newsletter.
Download PDF of this article:
unable to download the file
I have updated the article with the PDF file. Now you can download it by clicking on the download button.